
ERAD Therapeutics Inc.
- Location : Ontario, Canada and Delaware, USA
- Sector : Biotechnology

Growthbridge ventures is an Investor Relations and Marketing Outreach firm. The companies listed on this website and Portal are not an offer to the general public to purchase any form of securities. Every company listed on this website and Portal are listed for Investor Relations and marketing outreach, and should be considered to be a very high risk investment and the investor must be comfortable in accepting and bearing the very high level of risk associated with any of the companies listed on this site, should they choose to engage in any form of investment. The information contained on these companies is proprietary and strictly confidential. It is intended to be reviewed only by accredited investors and financial institutions and should not be used for any other reason or made available to any other person or entity without the prior written consent of Growthbridge ventures. The technology and process used on this website is protected by existing or pending patents owned by Growthbridge ventures. Nothing on this site including any related communication is intended to provide specific financial, investment, tax, legal, accounting or other advice to you, and should not be acted or relied upon in that regard without seeking the advice of a professional. In all circumstances, you should seek guidance from your current advisor as your advisor can help to ensure that your own circumstances have been properly considered and any action is taken on the latest available information.
ERAD Therapeutics Inc. (ETI) is an early-stage biotechnology company advancing proprietary therapeutic technologies for high-need, rare genetic diseases with limited or no current treatment options.
The company has been under new management and board leadership since 2023. ETI’s lead innovation, mCT, is a novel platform capable of addressing a wide range of rare genetic disorders associated with misfolded proteins and endoplasmic reticulum-associated degradation (ERAD).
The technology can be used alone or in combination with existing therapies to preserve protein function and improve disease outcomes. ETI also has access to proprietary gene therapy vectors targeting rare diseases and is exploring partnerships for broader applications in larger market indications.
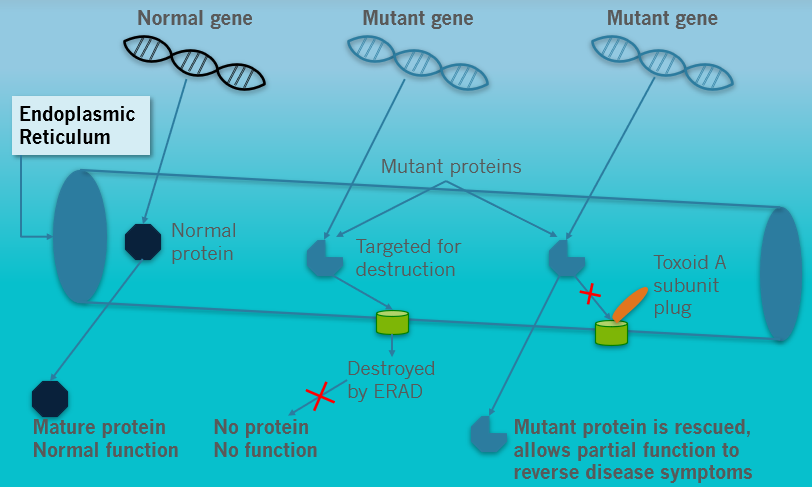
ERAD’s proprietary mCT technology temporarily halts the intracellular degradation of misfolded, partially functional proteins, thereby restoring normal—or near-normal—cellular activity.
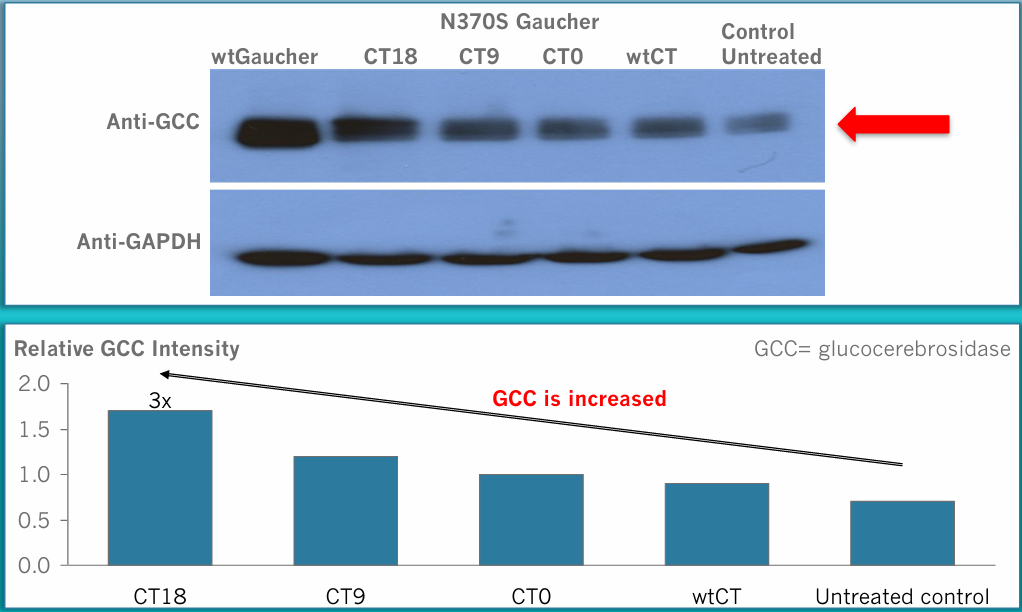
This therapeutic effect has been demonstrated in both cellular and animal models of Cystic Fibrosis, Gaucher Disease Type 1, Tay-Sachs Disease, and SP1 Lysase Deficiency. The company believes that mCT is a “gene-agnostic” platform with broad potential to treat a wide range of rare genetic disorders in which misfolded proteins retain partial function. It may also serve as a critical therapeutic bridge for ultra-rare diseases lacking viable treatment options, offering hope until more permanent solutions such as gene therapy become available.
ERAD is advancing its proprietary mCT platform through preclinical development, including toxicology studies slated for 2025, with plans to enter human clinical trials in 2026. Initial clinical efforts will focus on one to three ultra-rare genetic diseases where misfolded proteins retain partial function.
In parallel, the company has in-licensed a complementary gene therapy platform—complete with a robust toxicology dataset—and intends to commence animal model studies in 2025. The goal is to reach clinical development readiness for this gene therapy program by 2026.
Craig brings over 25 years of experience in early-stage drug development, with a focus on cancer and rare genetic diseases. With a background in biochemistry and life sciences, he has held research and commercial roles at Schering-Plough (Merck), Amgen, and Ares-Serono (Merck), contributing to the success of biologics like Intron-A®, Neupogen®, and Rebif®.
David has more than 30 years of pharmaceutical sales and marketing experience across the U.S., Canada, and international markets. His expertise spans discovery through commercial launch, with a track record of developing and executing strategic plans across diverse therapeutic areas.
Jeff is a veteran executive with 33 years in finance and operations for private and public life science companies. His experience includes investment banking, strategic transactions, M&A, licensing, and financial planning—backed by deep knowledge of corporate governance, IP, and regulatory frameworks.
Dr. Michaels is a physician executive with extensive leadership experience in advancing innovative treatments from discovery to market. She has guided programs from pre-IND through successful commercialization, with expertise in gene therapies, recombinant proteins, CRISPR editing, and pediatric rare diseases.
